
Hitachi Value Chain Traceability service for Regenerative Medicine
Regenerative Medicine Product Value Chain Integrated Management Platform: Birth of a Hitachi Value Chain Traceability Service for Regenerative Medicine
Hitachi, Ltd. has established a platform to manage trace information the entire value chain of regenerative medicine products, through co-creation with Alfresa Corporation, pharmaceutical companies, medical institutions, and other related parties.
This is Japan's first common service platform for regenerative medicine products, accessible to all stakeholders involved in the value chain (including medical institutions, pharmaceutical companies, logistics operators, and manufacturers).
The three key forms of value provided are as follows.
Through the provision of these forms of value, Hitachi will support the transformation of business processes in supply chain management, contributing to shorter lead times and the supply of high-quality regenerative medicine products.
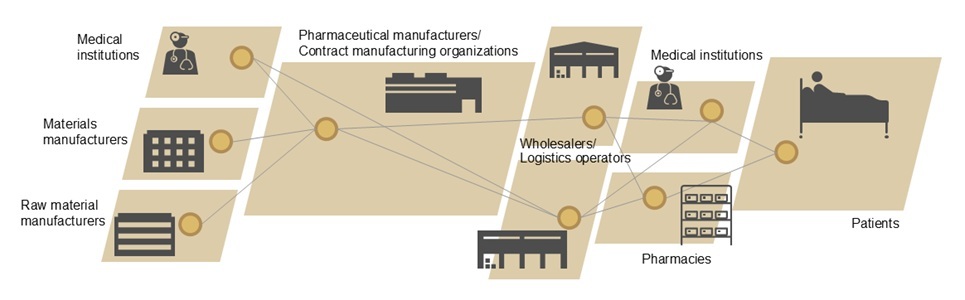
Pharmaceutical products are subject to strict management of all business processes and records within the supply chain, as their quality has a direct impact on lives.The distribution of regenerative medicine products including cellular medicines in particular requires traceability of information and individual tracking of cellular material and products throughout all processes. This is due to the characteristic nature of these products, which involve culturing cells collected from patients or cell donors and administering them to patients.
In view of these characteristics, Hitachi leverages its proven track record, technologies, and expertise to provide various production equipment, devices, and IT systems in the field of regenerative medicine products, offering a standard platform that supports the optimization of the entire supply chain, from cell collection and production to transportation and administration.
Collects and traces quality event information for each stakeholder on a unified platform
Individual identification: To link patient IDs and work instruction data in regenerative medicine projects at universities and medical institutions, we utilize the Patient Registry Service (*1) a service with an extensive proven track record in patient information management.
Information traceability: As our information infrastructure to trace quality information throughout the supply chain, including cell collection, production, transportation, and administration, we utilize Hitachi's proven IoT Compass (*2).
The service specifications aim for standard utilization by a broad range of stakeholders, based on considerations made in light of actual business processes, drawing on the expertise of Alfresa Corporation for aspects concerning the transportation and storage of patient cells and regenerative medicine products; contract manufacturing organizations for aspects concerning the development of manufacturing methods and the production of investigational drugs and regenerative medicine products; and pharmaceutical companies and biotech ventures for tracing overall process information from the collection of patient cells to the administration of regenerative medicine products.
To ensure efficient use at actual medical institutions, we made efforts such as incorporating feedback from healthcare professionals into the development of the user interface.

Managing schedule changes that occur for various reasons throughout the value chain from the planning stages of treatment and manufacturing, through cell collection, to the administration of regenerative medicine products is critical.
By sharing the status of each stakeholder, it is possible to comprehensively plan and modify optimal scheduling across the entire value chain and manage orders and shipments in a unified manner.
We also plan to expand functionality in the future, such as with the addition of automatic scheduling based on the actual results of plans.
*1 Launching "Patient Registry Services" to support the rapid development of pharmaceuticals and medical devices
@News releases: October 20, 2017: Hitachi
Hitachi has built a platform to manage tracability information the entire value chain of regenerative medicine products.
The platform is Japan's first common service platform for regenerative medicine products, accessible to all stakeholders involved in the value chain (including medical institutions, pharmaceutical companies, logistics operators, and manufacturers).
Related links
Related products
"HVCT RM is a trademark or registered trademark of Hitachi, Ltd. in Japan, China, and/or other countries."
iHVCT RMëAº{AA´Ìî^ɧë£ä¥äè´₤ÕÛÿźϣšäÊWɧëo^ÊWéñBj
